El fabricante del SW debe aplicar la norma IEC62366 para verificar requisitos de usabilidad

Clasificación del SW según MDR
La regla 11 del MDR y la guía MDCG2019-11 El MDR en su Anexo VIII
El fabricante del SW debe aplicar la norma IEC62366 para verificar requisitos de usabilidad

La regla 11 del MDR y la guía MDCG2019-11 El MDR en su Anexo VIII
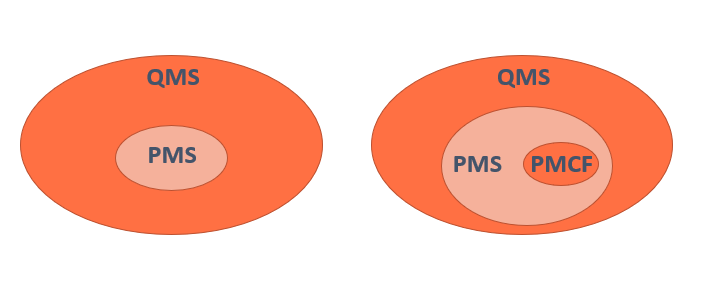
El MDR ha introducido nuevos requisitos para el Post Market Clinical Follow-up (PMCF). Introducción Según

Los fabricantes de Software como Producto Sanitario o SaMD (Software as Medical Device) tienen que
| Cookie | Duración | Descripción |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |